Limiting acetic acid accumulation during red wine aging with Hideki tannin and Stab Micro M (Enartis) (2023)
Lee Hartman
Bluestone Vineyards
Summary
Acetic acid accumulation during barrel aging is a significant concern for winemakers due to its impact on wine quality. This study evaluates the effectiveness of Hideki Tannin and Stab Micro M (chitosan) in controlling volatile acidity in red wines during aging. Cabernet Sauvignon and Cabernet Franc wines were monitored for acetic acid production, microbial populations, and oxidative changes. While Hideki Tannin did not significantly affect acetic acid accumulation or microbial load, sensory analysis suggested subtle shifts in aroma perception. Stab Micro M treatment resulted in slightly lower microbial populations but did not significantly reduce acetic acid levels. These findings suggest that while both treatments have potential as preventive measures, their effectiveness in controlling volatile acidity during barrel aging is limited when used alongside SO₂. However, their impact may be more pronounced in low-SO₂ applications. Additionally, since acetic acid levels were already relatively high at the start of aging, interventions during alcoholic and malolactic fermentation would likely be more effective.
Introduction
Acetic acid accumulation is one of the most common concerns for Virginia Winemakers. In addition to its own sensory impacts, volatile acidity can also decrease fruitiness and aromatic expression of the wine, reducing overall wine quality1. The aim of this experiment was to measure the concentration of acetic acid at various time points during wine production and aging, identify critical control points for prevention, and test two methods for reducing acetic acid accumulation during barrel aging.
Acetic acid accumulates due to the activity of microbes, including non-Saccharomyces yeast, stressed Saccharomyces, acetic acid bacteria, Brettanomyces bruxellensis, and others. Unfortunately, many of these microbes are ubiquitous on grapes or in wineries 2,3. Microbes need oxygen to produce acetic acid4, so limiting the microbial load and available oxygen are key strategies to limiting acetic acid production.
Most wineries have numerous operations in to prevent oxidation and limit the growth and activities of microbial populations. Some of the most common are:
- Gassing juice prior to inoculation to prevent surface microbes (Hanseniaspora) from proliferating and producing acetic acid.
- Providing adequate nutrition conditions that promote strong fermentation and prevents yeast stress (which results in acetic acid production).
- Regular post fermentation barrel topping to limit oxygen and prevent growth of acetic acid bacteria on surface layers.
- The addition of SO2 at the end of malolactic fermentation to provide both antimicrobial and antioxidant protection, if used in sufficient amounts.
- Judicious hygiene including cleaning and sanitizing tanks, hoses, barrels, fittings, and the pump, which limits biofilm formation and potential for cross inoculation.
However, these strategies are not always sufficient to prevent the accumulation of acetic acid above the level that impacts wine quality. Most textbooks4–6 list the threshold of detection for volatile acidity at or near 0.7 g/L, though values as low at 0.4 g/L have been shown to suppress fruitiness and oak character1. Values over 0.9 g/L imparting harsh, bitter or sour taste. When wine reaches 1.2 – 1.4 g/L (also the legal limit for white and red wines respectively), it may have a recognizable vinegar character.
In addition to oxygen exclusion and SO2 addition, chitosan and selected tannins have been developed to limit microbial activity during wine aging. Chitosan is a biopolymer used for its antimicrobial properties against Brettanomyces and other microbes. Different formulations of chitosan are commercially available that target different microbes. Though racking after treatment is recommended for wine with known microbial contamination, product information now also includes instructions for preventative treatment without racking. Previous WRE experiments using Stab Micro M on must at harvest (Lost Creek, 2021, Blenheim 2019) had mixed results. At Blenheim, there were no differences in acetic acid or microbial load on the treated wines while at Lost Creek (2021), wine treated with Stab Micro M finished fermentation with lower acetic acid and ethyl acetate, though these were not detected as different in a triangle test. In both experiments Stab Micro M was used during fermentation rather than during aging, as occurred at Bluestone.
Tannins are commonly added to wine to add structure and work as antioxidants. There is some evidence that selected phenolics compounds can also have antimicrobial properties. Phenolics are produced by plants as part of the defense mechanism against oxidation and microbial damage. Selected phenolic compounds have been shown to inactivate and even kill lactic acid bacteria in wine7–9, though other phenolic compounds may feed these microbes. Though the mechanism of action is not well understood, possible mechanisms include disruption to the cell membrane and binding to cell walls to block transport (much like chitosan).7–9
Hideki tannin, also sold by Enartis, works as both an antioxidant and an antimicrobial agent. A mix of gallic, ellagic and condensed tannins, Hideki is formulated to interfere with transport proteins of microorganisms and well as chelating metals that act as catalysts in oxidative pathways, and to quench free radicals produced by oxidation (Enartis technical sheet). In a previous WRE experiment (King Family, 2021), Hideki was one of three compounds tested to prevent malolactic fermentation in sparkling wine base. Though one of two barrels had signs of lactic acid depletion, that depletion may have occurred before Hideki was added. The Hideki treated wine was noticeably fresher to the winemaker and WRE Enologist post aging and was different in a triangle test.
Both chitosan and Hideki may be active against a wide range of microbes, including Oenococcus oeni, so addition occurred after the completion of malolactic fermentation (according to manufacturer instructions) to avoid interference with malic acid consumption.In this experiment, the production of acetic acid was monitored in two different lots of red wine at key points in fermentation and aging. At the end of malolactic fermentation, each 6-barrel lot was split into two treatments of three barrels each. Cabernet Sauvignon (3 barrels) was treated with 10 g/hL Hideki tannin, the maximum dose recommended, with three barrels as control. Cabernet Franc (3 barrels) was treated with 10 g/hL Stab Micro M. Because this is a prophylactic dose, wine was not racked post addition, but rather aged in the presence of chitosan.
See Appendix A for a broader review of chitosan use in the winery.
Cabernet Sauvignon/Hideki
Methods
Cabernet Sauvignon grapes were harvested on 10/8/23 and processed on 10/9/23. Grapes were destemmed to TBins with the addition of 40 ppm SO2 and 1 g/L tartaric acid. A juice sample for chemistry and microbial analysis was taken immediately after destemming on 10/9/23. A second sample for wine chemistry analysis was taken at the completion of malolactic fermentation on 2/19/24. The wine underwent co-inoculation for malolactic fermentation, so no intermediate chemistry samples were taken between alcoholic fermentation and malolactic fermentation. Post-pressing, 1 g/L of tartaric acid was added. At the completion of malolactic fermentation, 100 ppm SO2 was added to each barrel. The wine was not racked before aging, and barrels were topped monthly during the aging process.
Barrels were selected for Hideki tannin treatment to account for barrel-to-barrel variability. Notably, barrels 6 and 7 exhibited higher acetic acid concentrations than the other barrels. Barrels 3, 4, and 6 were designated as controls (with SO2 addition only), while barrels 2, 5, and 7 received 10 g/hL of Hideki tannin. The wine remained in barrel until December 2024, at which point additional chemistry and microbiological analysis was conducted.
Sensory analysis was completed by a panel of 31 wine producers using shipped samples. Every wine producer received 3 wines in identical bottles, filled on the same day, each coded with random numbers. Of the three wines, two were identical and the participant was asked to identify which wine was different (a triangle test). There were four tasting groups with the unique wine in the triangle test balanced between groups. Tasters were then asked to score each wine on a scale of 0 to 10 for color hue, fruit intensity, oxidation/reduction, tannin, and volatile acidity. They were also given open ended questions to describe the wines. Results for the triangle test were analyzed using a one-tailed Z test. Descriptive scores were analyzed using repeated measures ANOVA.
Results
Juice chemistry (Table 1) and microbiology (Table 4) indicate the grapes were ripe and healthy at the time of harvest. There was no detectable acetic acid. Hanseniaspora, a common non-Saccharomyces yeast found in grape must, has the potential to produce both beneficial aromas as well as high concentrations of acetic acid and ethyl acetate if oxygen is available10. This microbe was present in sufficient density to produce acetic acid and ethyl acetate, but on the low end of this range. Though exact density values are hard to find, generally, cells need to be present at a density of 104 cells/L to produce spoilage, with higher density usually found in spoiled wines.
Table 1: Juice Chemistry (Imbibe Solutions, 10/9/23)

All the wines finished fermentation with glucose/fructose < 1.0 g/L and malic acid <0.15 g/L (data not shown). Ethyl acetate values were below the threshold of detection (90-150 mg/L) (Table 2). However, acetic acid values were already near (barrels 2-5) or above (barrels 6 & 7) the reported threshold for detection (0.7 g/L11) . These values are still below threshold for rejection (0.9 g/L11), so limiting additional accumulation during aging was very important.
Table 2: Wine chemistry at the completion of ML (ICV Labs, 2/19/24)
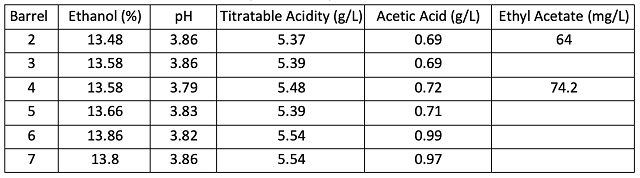
Hideki tannin is formulated to have both an antioxidant and antimicrobial benefits. Despite barrel to barrel variation, there was no difference in chemical measures of oxidation after aging (Table 3). The average acetic acid (0.92 g/L vs. 0.91 g/L) and color hue (0.8), a potential measure of oxidation over time, are the same between treatments. The average change in acetic acid during aging was identical for each treatment (0.12 g/L)(Tables 2 & 3). This accumulation is only slightly higher than reported values due to chemical oxidation alone (0.06 – 0.12 g/L/year)6. The slightly higher color intensity in the control wines (9.91 vs. 9.76) is likely due to slightly lower free SO2 values (27 vs. 29 ppm) in the Hideki treated wine rather than the impact of Hideki itself. These differences are not large and would not lead to any change in winemaking operations.
Table 3: Wine chemistry at the end of aging (ICV Labs, 12/16/24)
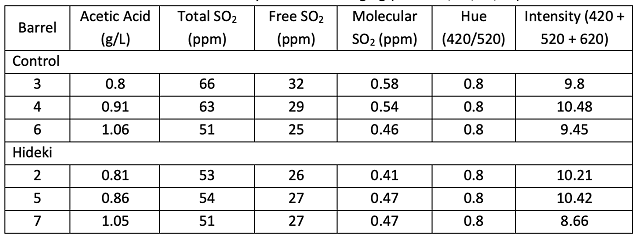
To better understand the potential antimicrobial impact of Hideki, a wine sample was taken in December after stirring, then sent to ETS labs for Scorpions analysis. Scorpions analysis detects DNA from living and dead microbes, so this analysis measures the accumulation of microbial populations over time rather than those that are currently active. The microbial community was also sampled at harvest. Those data are included here for comparison (Table 4).
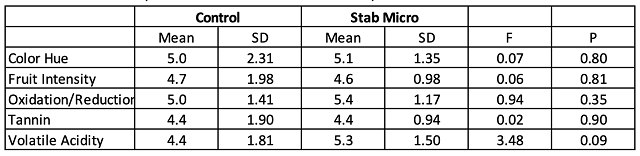
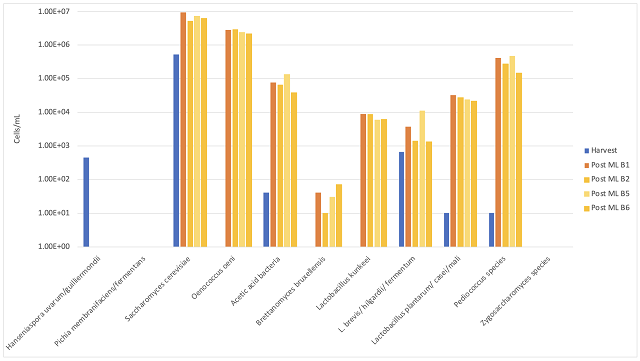
A large increase in Saccharomyces and Oenococcus population is the normal result of robust fermentation, and do not pose any danger of microbial contamination as long as all sugar and malic acid have been consumed (as was the case here). There is large barrel to barrel variation in several microbial groups, most notably the acetic acid bacteria and some Lactobacillae, but none of these differences correlates with Hideki treatment (Figure 1). The differences are more likely due to differences in wine population upon filling (for example, barrels filled last contain more lees from the settling tank), inoculum through the barrel itself, or differences in topping efficacy. (Acetic acid bacteria proliferate quickly in oxygen rich headspace.)
In this experiment, Hideki treatment did not lead to any measurable differences in oxidation (color hue, color intensity, SO2 depletion or acetic acid accumulation) nor microbial population size. However, the microbial population may have already been present post ML, and there was little overall oxidation to combat.
Table 4: Scorpions of yeast and bacteria for juice and wine. Hanseniaspora and Pichia were only tested in juice. (ETS Labs, 10/10/23 and 12/16/24)
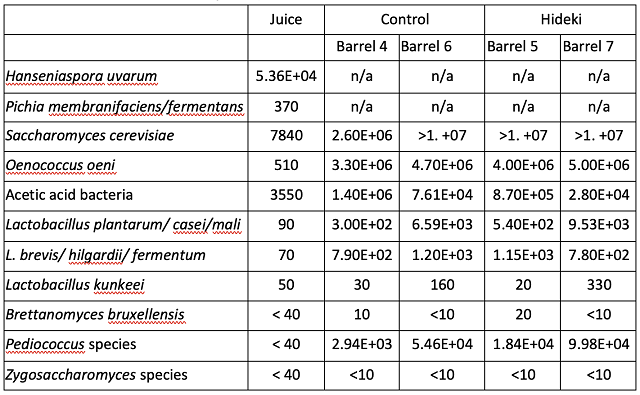
In a triangle test of Cabernet Sauvignon aged with and without Hideki tannins, 20 of the 32 respondents were able to distinguish which wine was different, indicating the wines were significantly different (Z=3.31, p=0.0005). However, there were no significant differences in scores for color hue, fruit intensity, oxidation/reduction, tannin, and volatile acidity (Table 5). Open-ended questions indicate that the tasters felt the wines aged without Hideki tannins had a more pronounced oak character, whereas the wines aged with Hideki tannins were more fruit forward and had notes of warm spices and vanilla. The wine was aged in comparable neutral barrels, so differences in oak character were not likely due to oak impact itself. These differences were more likely due to shifts in the perception of aroma vectors for fruit and spice, potentially due to less oxidation.
Table 5: Repeated Measures ANOVA of descriptive scores in Cabernet Sauvignon
Cabernet Franc/Stab Micro
Methods
Cabernet Franc grapes were harvested on 10/10/23 and processed the following day, 10/11/23. Grapes were destemmed to TBins with the addition of 40 ppm SO2 and 1 g/L tartaric acid. A sample for juice chemistry analysis was taken after destemming on 10/12/23, and wine chemistry analysis was performed at the end of malolactic fermentation on 2/19/24. The wine was co-inoculated for malolactic fermentation, so no chemistry data was taken between the end of alcoholic fermentation and the start of malolactic fermentation. However, at pressing, the pH was found to be high, prompting the addition of 1 g/L tartaric acid at barrel down. Once malolactic fermentation was complete, an additional 0.5 g/L tartaric acid was added, along with 100 ppm SO2. The wine was not racked before aging, and barrels were topped monthly during the aging process.
Odd-numbered barrels (1, 3, and 5) were kept as controls, receiving only 100 ppm SO2, while even-numbered barrels (2, 4, and 6) were treated with 100 ppm SO2 and 10 g/hL Stab Micro M*. Barrels 5 and 6 exhibited higher post malolactic acetic acid concentrations compared to the others; these were split up between treatments. The wine remained in barrel until December 2024, when further analysis of both wine chemistry and microbiology was conducted.
Sensory analysis was completed by a panel of 31 wine producers using shipped samples. Every participant received 3 wines in identical bottles, filled on the same day, each coded with random numbers. Of the three wines, two were identical and the participant was asked to identify which wine was different (a triangle test). There were four tasting groups with the unique wine in the triangle test balanced between groups. Tasters were then asked to score each wine on a scale of 0 to 10 for color hue, fruit intensity, oxidation/reduction, tannin, and volatile acidity. They were also given open ended questions to describe the wines. Results for the triangle test were analyzed using a one-tailed Z test. Descriptive scores were analyzed using repeated measures ANOVA.
* Enartis produces two different chitosan products, Stab Micro M and Stab Micro. Enartis recommends using Stab Micro M in turbid juice/must/wine while Stab Micro is recommended for use in clarified wine. Stab Micro M was used here, at the recommendation of the manufacturer, because the wine had recently finished fermentation and had not been racked.
Results
Juice chemistry (Table 6) and microbiology (Table 9, Figure 4) indicate the grapes were ripe and healthy at the time of harvest. There was no detectable acetic acid and very little Hanseniaspora or acetic acid bacteria.
Table 6: Juice chemistry (ETS Labs, 10/12/23)

All wines finished fermentation with glucose/fructose < 1.0 g/L and malic acid <0.15 g/L). However, acetic acid values were already near (barrels 1-4) or above (barrels 5 & 6) the reported threshold for detection (0.7 g/L11) (Table 7). These values are still below the threshold for rejection (0.9 g/L11), so limiting any additional accumulation of acetic acid during aging was very important.
Table 7: Wine chemistry at the completion of ML (ICV Labs, 2/19/24)
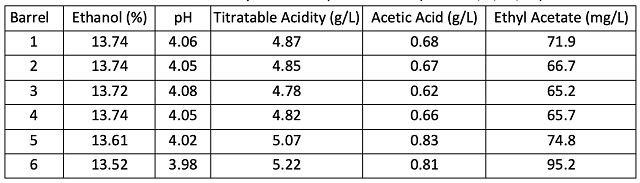
All barrels had some accumulation of acetic acid during aging; however, the average amount was relatively low, and nearly the same for both treatments (0.12 and 0.13 g/L, respectively) (Tables 7 &8).
Microbial analysis of the wine after stirring in April 2024 and December 2024 are shown in Tables 9 & 10. As seen in the Cabernet Sauvignon experiment, large populations of Saccharomyces and Oenococcus are the normal result of robust fermentation, and do not pose any danger of microbial contamination if all sugar and malic acid have been consumed (as is the case here). Once again, there is large barrel to barrel variation in microbial load, however, when compared to initial values, there is an overall pattern of lower microbial population growth in the barrels treated with Stab Micro M compared to control (Table 10, Figure 2). Though most of the barrels still contained a sufficient density of microbes (>104 cells/mL) to produce acetic acid if suitable conditions arose (such as the introduction of oxygen and relief from SO2), some of these cells may not be active. Here, Stab Micro M treatment led to slightly lower microbial population when used in a prophylactic dose without racking. Nardi at al (2014)12 found a 100-fold decrease in the population density of Brettanomyces with application of a prophylactic dose of chitosan, with persistent effects for 180 days. The impact here was not as profound, however, chitosan treated barrels on average had lower microbial load after treatment than before.
Table 8: Wine chemistry at the end of aging (ICV Labs, 12/16/24)
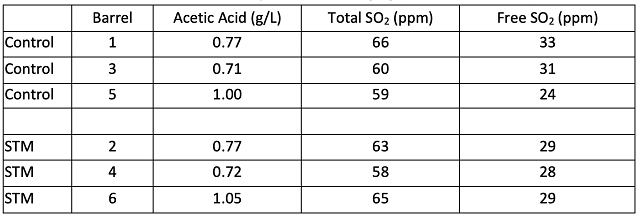
Table 9: Microbiology from juice to wine (ETS labs, 10/12/23 & 2/19/24)
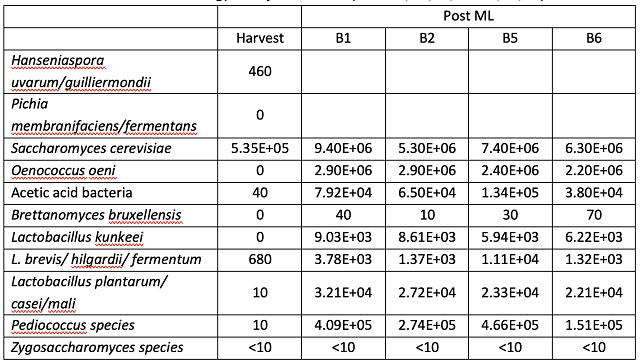
Table 10: Post treatment and aging microbiology ETS labs, 12/16/24)
In a triangle test of Cabernet Franc aged with and without Stab Micro, 7 of the 31 respondents were able to distinguish which wine was different, indicating that the wines were not statistically significant (Z=-1.46, p=0.07). If p value restrictions were relaxed to 0.1, the wine aged with Stab Micro scored significantly higher in scores for volatile acidity than the control wine. There were no significant differences in scores for color hue, fruit intensity, oxidation/reduction, or tannin (Table 11).
Table 11: Repeated measures ANOVA of descriptive scores for Cabernet Franc
Overall acetic acid accumulation
With or without Hideki or Stab Micro M, there was very low overall accumulation of acetic acid in these wines after malolactic fermentation. Zoecklein (1995) states that 0.06 - 0.12 g/L/year of acetic acid accumulation is expected during barrel aging due to chemical oxidation alone6. At Bluestone, a strong addition of SO2 coupled with monthly topping kept this accumulation to an average of 0.18 g/L/year. However, these wines were still at or near the limit of detection for volatile acidity due to accumulation during alcoholic and malolactic fermentation (Figure 3).
Several elements of these wines may have put them at risk for microbial spoilage early on.
- The pH values post ML were at or above 3.8. The fruit came in with high pH (3.75, 3.82), and, even with acid addition, finished with high pH. High pH provides a more hospitable environment for spoilage organisms, so these wines may have benefited from more aggressive acid addition at the beginning of fermentation.
- Despite co-inoculation, the wines did not finish malolactic fermentation for several months, leaving the wines unprotected by SO2 until February. Typically, some headspace forms during ML as wine loses CO2 and wine degasses. Prolonged periods with no SO2 protection and ample oxygen in the headspace allows for proliferation and activity of acetic acid bacteria. Lactic acid bacteria other than Oenococcus can also produce acetic acid if malic acid and oxygen are available. Co-inoculation is usually done to hasten the end of ML; however these bacteria are sensitive to temperature and other conditions of fermentation. Additionally, if alcoholic fermentation stalls, Oenococcus can consume sugar to produce acetic acid.
In both wines, two of the 6 barrels have notably higher acetic acid values very early, indicating some possible barrel contamination. Wine from four of the Cab Franc barrels was sent for microbial analysis in April to determine if higher post malolactic acetic acid level in barrels 5 and 6 was due to higher microbial load. Though barrel 5 (a high acetic acid barrel) had higher density of acetic acid bacteria and L. brevis, the values for these microbes (and all others) were not elevated in barrel 6 relative to the other barrels (1 & 2). The production of acetic acid requires both the availability of oxygen as well as microbial metabolism. All the barrels had high enough density of acetic acid bacteria (104 cells/mL) for the production of acetic acid, so perhaps the difference in production was due to oxygen availability.
Figure 1: Comparison of the microbial population in control vs. Hideki treated Cabernet Sauvignon after aging 8 months post treatment (ETS Labs). Values from control barrels are shown in orange while treated barrels are shown in blue.
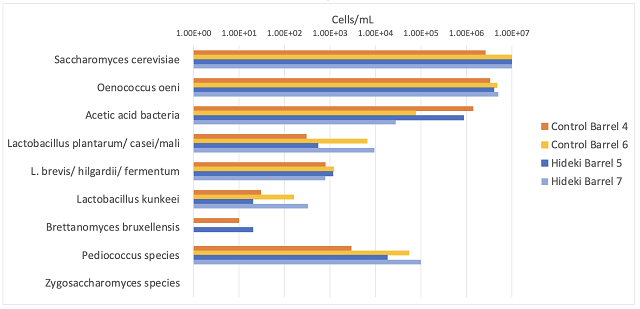
Figure 2: Comparison of the microbial population in control vs. Stab Micro M treated Cabernet Franc after aging 8 months post treatment (ETS Labs). Each bar represents the average value for two barrels per treatment. The initial population was measured post ML, in April 2023.
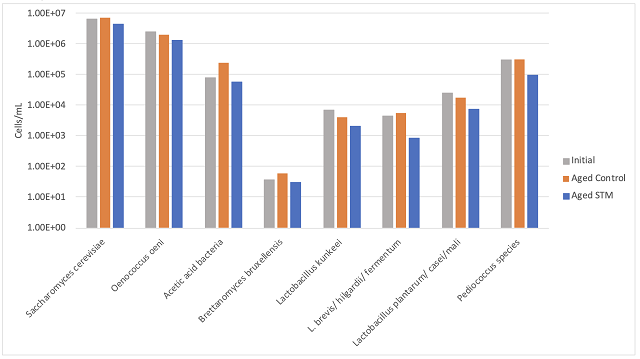
Figure 3: Accumulation of acetic acid in Cabernet Sauvignon (a) and Cabernet Franc (b) from harvest to bottling.
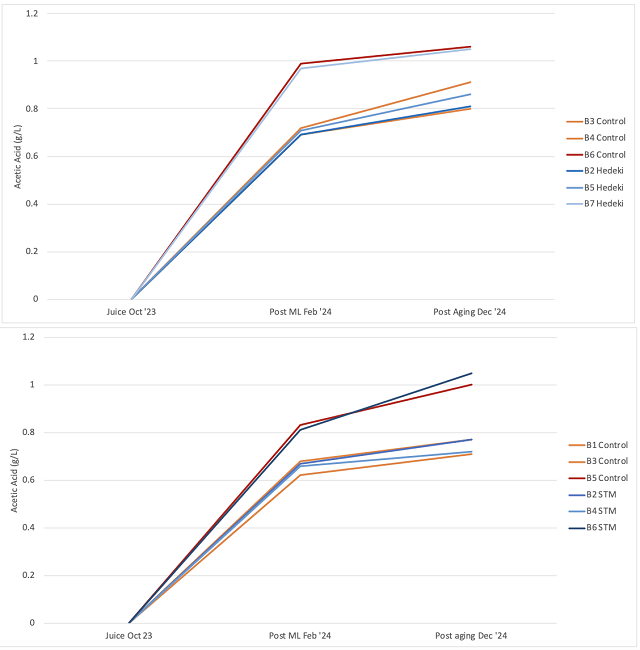
Figure 4: Growth in the microbial population from harvest through ML in four barrels of Cabernet Franc (ETS Labs, April 2024)
Appendix A: The mechanism and breadth of chitosan use in the winery.
Chitosan is a naturally occurring molecule that can also be produced by the de-acetylation of chitin using NaOH or chitinase enzymes13. The effectiveness of any given formulation of chitosan depends on its molecular weight, deacetylation degree and the pH of the medium14. Lower molecular weight, higher degree of deacetylation is the favored formulation for antimicrobial purposes15. At juice and wine pH, chitosan is very positively charged, which increases efficacy15. Chitosan binding to cell walls of microbes is driven by the chemical properties of the cell wall itself, with a high degree of correlation to the hydrophilicity of the wall16. Gram negative bacteria are more susceptible to binding than gram positive16. Chitosan has been shown to have some efficacy against a wide range of grapevine and wine microbes including downy mildew17, powdery mildew18, Phomopsis19, Lactobacillus, Oenococcus, and Brettanomyces16. Due to its versatility as an antimicrobial agent, chitosan in various forms has been used worldwide at nearly every stage of wine production including vineyard applications, on grapes during transport and storage, at crush, after fermentation and during the aging of wine15,20,21.
Many different mechanisms of antimicrobial action have been proposed for chitosan in wine. Chitosan has been shown to physically bind to the cell walls of microbes14,16. Binding may aid in sedimentation, leading to a reduction in the overall cell number with racking. Binding of the positively charged molecule of chitosan may also disrupt cell membranes, leading to leakage of ATP, potassium, and proteins, all essential components of cell function14,16. Therefore, leakage may result in semi-viable cells or cell death. Other proposed mechanisms include the physical blocking of cell permeability by chitosan binding, chelation of survival factors such as copper, and binding to the DNA after penetrating the cell membrane14.
Regardless of the mechanism, chitosan has been shown to be quite effective in treating microbial infection of wine. When treating wine already inoculated with Brettanomyces bruxellensis, Taillander et al (2014)14 found that 85% of the population of Brettanomyces cells were dead within 20 hours of treatment. There was a dose effect in the rate of cell death, with 0.4 g/L treatment killing cells faster than 0.04 g/L. They also found differences in susceptibility based on the strain of Brettanomyces used, presumably due to genetic differences in cell wall components. Many cells initially compromised by chitosan treatment recovered, as evidenced by growth in the Brettanomyces population after 7 days. Other researchers tested the effect of 0.04 g/L chitosan on aging wine and found that, even at low dose, aging of wine on chitosan helped prevent infection by Brettanomyces22.
References
(1) Ferreira, V.; De La Fuente, A.; Sáenz-Navajas, M. P. Wine Aroma Vectors and Sensory Attributes. In Managing Wine Quality; Elsevier, 2022; pp 3–39.
(2) Joyeux, A.; Lafon-Lafourcade, S.; Ribéreau-Gayon, P. Evolution of Acetic Acid Bacteria During Fermentation and Storage of Wine. Appl Environ Microbiol 1984, 48 (1), 153–156.
(3) Bokulich, N. A.; Thorngate, J. H.; Richardson, P. M.; Mills, D. A. PNAS Plus: From the Cover: Microbial Biogeography of Wine Grapes Is Conditioned by Cultivar, Vintage, and Climate. Proceedings of the National Academy of Sciences 2014, 111 (1), E139–E148.
(4) Ribereau-Gayon, P.; Glories, Y.; Maujean, A.; Dubourdieu, D. Handbook of Enology Volume 2: The Chemistry of Wine Stabilization and Treatments, 2nd ed.; John Wiley & Sons, Ltd: West Sussex, England, 2006; Vol. 2.
(5) Jackson, R. S. Wine Science: Principles and Applications, 4 edition.; Academic Press: Amsterdam, 2014.
(6) Zoecklein, B.; Fugelsang, K. C.; Gump, B. H.; Nury, F. S. Wine Analysis and Production; Springer: New York, 1995.
(7) García-Ruiz, A.; Moreno-Arribas, M. V.; Martín-Álvarez, P. J.; Bartolomé, B. Comparative Study of the Inhibitory Effects of Wine Polyphenols on the Growth of Enological Lactic Acid Bacteria. International Journal of Food Microbiology 2011, 145 (2–3), 426–431.
(8) García-Ruiz, A.; Bartolomé, B.; Cueva, C.; Martín-Álvarez, P. J.; Moreno-Arribas, M. V. Inactivation of Oenological Lactic Acid Bacteria ( Lactobacillus Hilgardii and Pediococcus Pentosaceus ) by Wine Phenolic Compounds. Journal of Applied Microbiology 2009, 107 (3), 1042–1053.
(9) García-Ruiz, A.; Bartolomé, B.; Martínez-Rodríguez, A. J.; Pueyo, E.; Martín-Álvarez, P. J.; Moreno-Arribas, M. V. Potential of Phenolic Compounds for Controlling Lactic Acid Bacteria Growth in Wine. Food Control 2008, 19 (9), 835–841.
(10) Jolly, N. P.; Varela, C.; Pretorius, I. S. Not Your Ordinary Yeast: Non- Saccharomyces Yeasts in Wine Production Uncovered. FEMS Yeast Res 2014, 14 (2), 215–237.
(11) Ask the AWRI: Volatile Acidity. Grapegrower and Winemaker 2018, No. 648.
(12) Nardi, T.; Vagnoli, P.; Minacci, A.; Sauthier, S.; Sieczkowski, N. Evaluation of the Impact of a Fungal-Origin Chitosan Preparation on Brettanomyces in the Context of Wine Aging; Beune, France, 2014.
(13) O’Kennedy, K. Chitin, chitinase, chitosan ... Wineland Magazine. https://www.wineland.co.za/chitin-chitinase-chitosan/ (accessed 2019-10-18).
(14) Taillandier, P.; Joannis‐Cassan, C.; Jentzer, J.-B.; Gautier, S.; Sieczkowski, N.; Granes, D.; Brandam, C. Effect of a Fungal Chitosan Preparation on Brettanomyces Bruxellensis, a Wine Contaminant. Journal of Applied Microbiology 2015, 118 (1), 123–131.
(15) Barrett, L. Microbial Stability and Control: EnartisStab Micro (Chitosan) Application during Wine Maturation, 2019. https://www.enartis.com/en-us/video-e-webinar/.
(16) Chung, Y.; Su, Y.; Chen, C.; Jia, G.; Wang, H.; Wu, J. C. G.; Lin, J. Relationship between Antibacterial Activity of Chitosan and Surface Characteristics of Cell Wall. Acta Pharmacol Sin 2004, 5.
(17) Romanazzi, G.; Mancini, V.; Feliziani, E.; Servili, A.; Endeshaw, S.; Neri, D. Impact of Alternative Fungicides on Grape Downy Mildew Control and Vine Growth and Development. Plant Disease 2016, 100 (4), 739–748.
(18) Iriti, M.; Vitalini, S.; Di Tommaso, G.; D’Amico, S.; Borgo, M.; Faoro, F. New Chitosan Formulation Prevents Grapevine Powdery Mildew Infection and Improves Polyphenol Content and Free Radical Scavenging Activity of Grape and Wine: Grape Powdery Mildew Control by Chitosan. Australian Journal of Grape and Wine Research 2011, 17 (2), 263–269.
(19) Nascimento, T.; Rego, C.; Oliveira, H. Potential Use of Chitosan in the Control of Grapevine Trunk Diseases. Phytopathologia Mediterranea 2007, 46 (2), 8.
(20) Singh, R. K.; Martins, V.; Soares, B.; Castro, I.; Falco, V. Chitosan Application in Vineyards (Vitis Vinifera L. Cv. Tinto Cão) Induces Accumulation of Anthocyanins and Other Phenolics in Berries, Mediated by Modifications in the Transcription of Secondary Metabolism Genes. IJMS 2020, 21 (1), 306.
(21) Singh; Soares; Goufo; Castro; Cosme; Pinto-Sintra; Inês; Oliveira; Falco. Chitosan Upregulates the Genes of the ROS Pathway and Enhances the Antioxidant Potential of Grape (Vitis Vinifera L. ‘Touriga Franca’ and ’Tinto Cão’) Tissues. Antioxidants 2019, 8 (11), 525.
(22) Bağder Elmaci, S.; Gülgör, G.; Tokatli, M.; Erten, H.; İşci, A.; Özçelik, F. Effectiveness of Chitosan against Wine-Related Microorganisms. Antonie Van Leeuwenhoek 2015, 107 (3), 675–686.
